COVID-19 치료제개발 글로벌 임상연구 프로젝트현황
신종 코로나바이러스 감염증(코로나19) 치료제 개발을 위해 전세계가 노력하고 있는 가운데, 현재 코로나 19 치료제 개발과 관련 국제적으로 임상연구가 335,815건이 진행 중에 있다(2020년 4월 13일자 등록현황). 미국 국립보건원 NIH ClinicalTrials.gov에 등록되어 진행하고 있는 COVID-19 관련 임상연구는 300여건 이상이 진행 중에 있으며, 세계보건기구(WHO)국제임상연구등록 플랫폼(World Health Organization's International Clinical Trials Registry Platform, WHO ICTRP)에서는 590건의 프로젝트가 현재 임상연구 중에 있다.
ClinicalTrials.gov: Federally-funded clinical studies related to COVID-19
WHO Trial Registry Network: COVID-19 studies from the ICTRP database
CDC: Information for Clinicians on Therapeutic Options for COVID-19 Patients

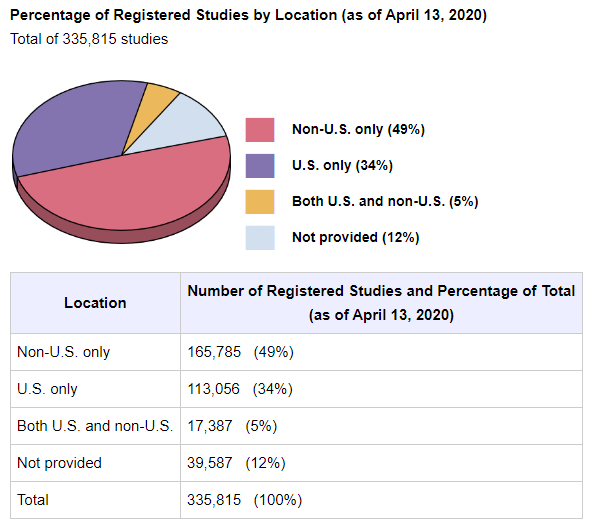
국가별 지역별로 진행중인 연구를 살펴보면 전체의 미국이 34%를 차지하고 있으며 미국 이외에서 수행되는 임상연구는 49%로 아래 그림과 같다.
연도별로 ClinicalTrials.gov에 등록된 임상연구 현황을 살펴보면 아래와 같다.
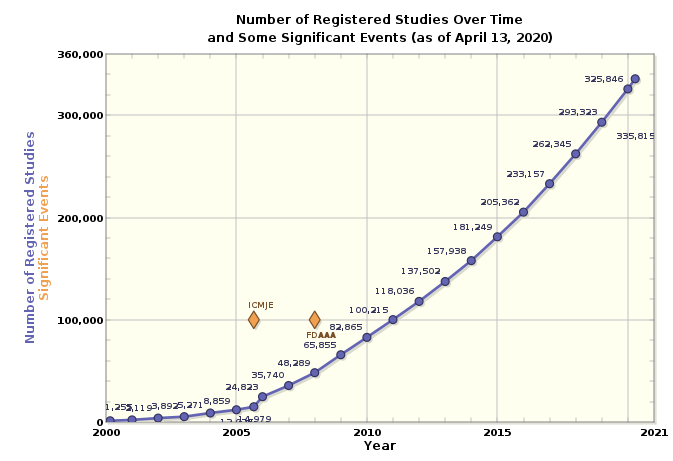
Source: http://clinicalTrials.gov


[원문바로가기]

의과학연구정보센터 제공